Enabling Safe and Effective Regulation of Genome Editing in Africa
POLICY BRIEF 2
Enabling Safe and Effective Regulation of Genome Editing in Africa
Target Audience: Regulatory Agencies and Biosafety Authorities
Gene editing (GEd) is making steady inroads into Africa’s research and innovation ecosystems, particularly in agriculture and health. As scientists explore its potential to address urgent challenges—such as crop resilience, disease control, and local production of biopharmaceuticals—regulatory systems must evolve in step with the technology’s growth.
GEd offers precision and flexibility in modifying the genetic material of organisms for beneficial purposes. However, its novelty and complexity present challenges for existing biosafety structures, especially where mandates, capacity, and legal provisions are still emerging or unclear.
The AUDA-NEPAD Gene Editing Landscape Study, covering 16 African countries, revealed significant disparities in regulatory preparedness. While some countries are developing or implementing GEd-specific guidelines, others continue to operate without defined regulatory pathways. For GEd to be safely and equitably deployed, national regulatory systems must be clear, science-informed, and forward-looking.
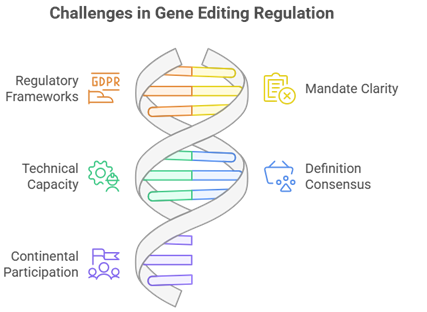
Key Findings from the Landscape Study
- Emerging GEd-Specific Guidelines: Only 6 of the 16 countries assessed had developed or adopted regulatory frameworks explicitly tailored to gene editing technologies, reflecting the early stage of policy development in this area.
- Fragmented Regulatory Mandates: Oversight responsibilities are often split across ministries or remain unclear within existing biosafety institutions, hindering coordinated governance.
- Insufficient Technical Capacity: Many regulatory bodies lack exposure to gene editing technologies, modern risk assessment tools, and structured opportunities for professional development.
- Divergent Interpretations and Definitions: Lack of consensus on how to define and categorize gene-edited products creates delays and uncertainty in implementation.
- Limited Participation in Continental Platforms: Several national regulators are not actively engaged in AU-level harmonization efforts or regional knowledge-sharing initiatives that could accelerate alignment.
Opportunities for Regulatory Leadership
Regulatory agencies play a gatekeeping role in ensuring the safe and responsible use of gene editing technologies. They are uniquely positioned to shape national and regional standards, foster public confidence, and support innovation aligned with national priorities. Strategic opportunities for leadership include:
- Adopting science-based, risk-proportionate frameworks that reflect the specific characteristics and use cases of gene editing.
- Strengthening cross-border collaboration to align terminology, risk categories, and regulatory procedures.
- Building internal technical capacity in areas such as genome editing risk analysis, regulatory review, and product evaluation.
- Engaging proactively with researchers and developers to ensure early dialogue, promote transparency, and anticipate emerging needs.
- Participating actively in continental platforms led by AUDA-NEPAD and other regional bodies to support regulatory harmonization and shared learning.
Policy Recommendations
- Review and revise national biosafety laws to provide clear legal pathways for the regulation of gene editing technologies, including definitions, scope, and procedural guidance.
- Clarify the mandates and coordination mechanisms of regulatory authorities to avoid overlaps, strengthen accountability, and ensure coherent oversight across sectors.
- Invest in regulator-specific capacity development through targeted training, exchange programmes, and technical support on gene editing science, risk assessment, and regulatory decision-making.
- Engage in continental and regional harmonization efforts to align definitions, standards, and approval processes, and to facilitate mutual recognition across borders.
- Promote inclusive, transparent regulatory processes by involving scientists, innovators, and the public in structured consultations and communication strategies.
Call to Action
As gene editing technologies continue to expand across Africa, the role of regulatory agencies is pivotal. Regulatory systems that are forward-looking, well-resourced, and regionally connected will be essential to ensure safe, responsible, and equitable deployment—while also enabling countries to meet their innovation and development goals.
AUDA-NEPAD stands ready to support national regulators through targeted technical assistance, capacity-building initiatives, and facilitation of continental coordination and policy alignment.
Contact: AUDA-NEPAD [Insert email/contact point]
